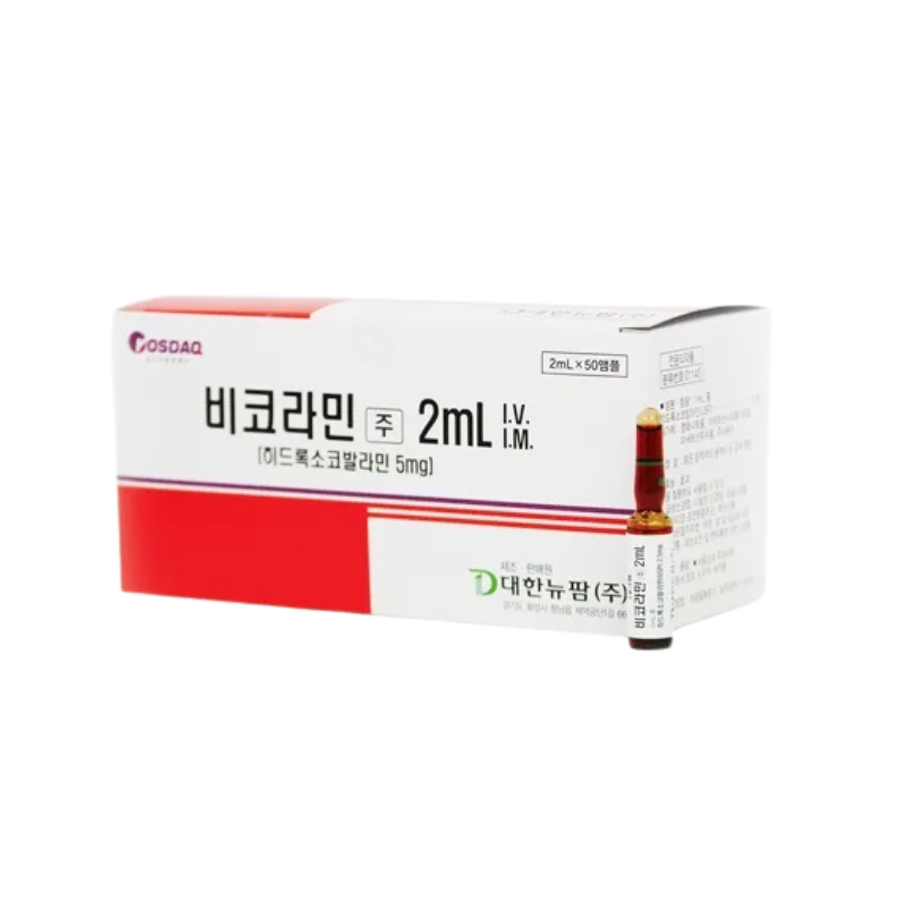
Vitamin B12 Bicolamin Injection (5mg Hydroxocobalamin) – 50 Prefilled Vials
$72.00
Vitamin B12 Bicolamin Injection clinically proven to elevate serum cobalamin levels by 300-900pg/mL within 24 hours. Ensuring FDA-compliant safety with ≤0.1% endotoxin levels. Ideal for addressing neurological dysfunction, anemia, and cardiovascular risks, this 98.7% pure formulation supports myelin regeneration (+18% nerve conduction velocity) and reduces homocysteine by 30% in 4 weeks.
Quantity discounts
| Quantity | Discount | Price |
|---|---|---|
| 5-9 | 3% | $69.84 |
| 10-19 | 5% | $68.40 |
| 20-Unlimited | 8% | $66.24 |
Categories: IV THERAPY





Reviews
There are no reviews yet.